Polarity, shape and intermolecular forces 3/26
Urea is a polar covalent molecule because of the intramolecular bonds between the elements in Urea. The electrons are most attracted to Oxygen since it has the largest element of all; oxygen is therefore the most electronegative, having the negative end of the molecular bond. Also, Oxygen and Carbon has a double bond. Carbon is the central atom since it has the need to bond its four valence electrons to form its full octet. However, Nitrogen is more electronegative than Carbon since Nitrogen has a larger mass. Nitrogen has 3 valence electrons so it bonds with Carbon and Hydrogen to fill its outer shell. Hydrogen is the least electronegative therefore it has the positive end. It exists as a diatomic element but in molecules, it has one valence electron therefore bonding with the Nitrogen.
All of the electron activity and intramolecular bonds within Urea create a molecular shape of three trigonal planars: the first one is between carbon, nitrogen and oxygen while the other two are between Carbon Nitrogen and hydrogen. On the other side, the electroactivity within Urea heavily influence its interactions with other molecules and elements. Urea's intermolecular forces include dipole dipole forces upon the opposite end poles and hydrogen bonding due to the hydrogens bonded with nitrogen. Because hydrogen bonding is between the hydrogen of a molecule and water, Urea is very solubule in water. Additionally, Urea is synthesized by the reaction of ammonia and carbon oxide. Urea also has a carboxyl group.
All of the electron activity and intramolecular bonds within Urea create a molecular shape of three trigonal planars: the first one is between carbon, nitrogen and oxygen while the other two are between Carbon Nitrogen and hydrogen. On the other side, the electroactivity within Urea heavily influence its interactions with other molecules and elements. Urea's intermolecular forces include dipole dipole forces upon the opposite end poles and hydrogen bonding due to the hydrogens bonded with nitrogen. Because hydrogen bonding is between the hydrogen of a molecule and water, Urea is very solubule in water. Additionally, Urea is synthesized by the reaction of ammonia and carbon oxide. Urea also has a carboxyl group.
Chemical Reactions with Urea
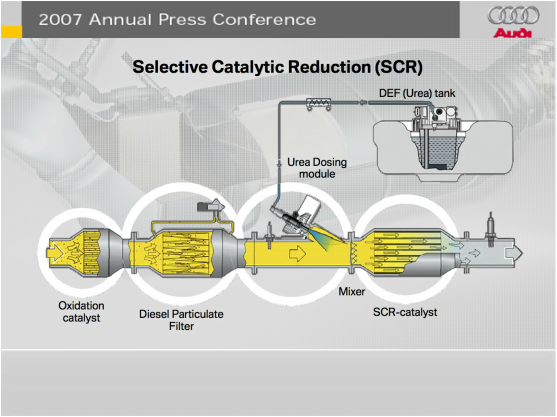
Urea in Car Engines |
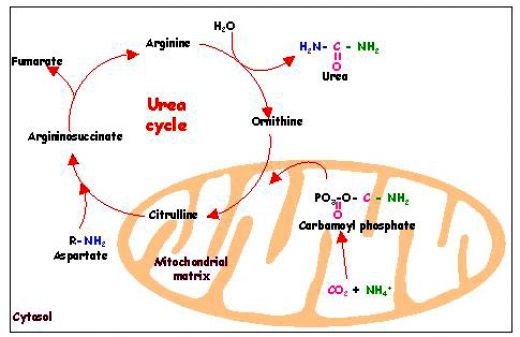
Urea Cycle |
|
Urea can be used in diesel fueled vehicles to reduce air pollution by converting the Nitrogen Oxide production from the exhaust emissions o Nitrogen and water. Urea is sprayed in the engine system's selective catalyst converter to let Urea separate nitrogen oxide to a green waste of nitrogen and water.
|
The Urea Cycle is used to rid the bodily production of ammonia by converting it into urea. The reaction occurs in the liver where its product, urea, gets transported to the kidney which will be excreted as urine.
Chemical Formula: Urea Cycle Disorders:
********* Symptoms include neonatal seizures. |
Urea Nitrate
Urea Nitrate is used as an explosive where nitric acid and urea are reacted together. It produces carbon dioxide, water and nitrogend oxide. Nitric Acid ionizes with the carbonyl group thus creating the ion, Urea Nitrate.
Chemical Formula: (NH2)2CO (aq) + HNO3 (aq) → (NH2)2COHNO3 (s)
Chemical Formula: (NH2)2CO (aq) + HNO3 (aq) → (NH2)2COHNO3 (s)
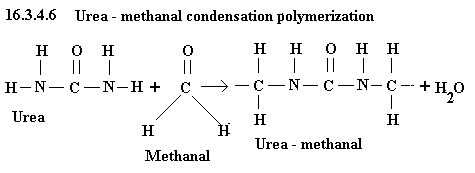
Urea as a limiting reactant
CH2O + CH4N2O = C3H7N2O + H2O
methanal + urea = urea methanal + water
200 g methanal * 1 mole methanal * 1 mole urea methanal = 6.65 mole urea methanal
30.031 g methanal 1 mole methanal
200g urea * 1 mole urea * 1 mole urea methanal = 3.33 mole urea methanal
60.06 g urea 1 mole urea
Conclusion:
Urea is the limiting reactant in this reaction since it produced the least product which is urea methanal.
methanal + urea = urea methanal + water
200 g methanal * 1 mole methanal * 1 mole urea methanal = 6.65 mole urea methanal
30.031 g methanal 1 mole methanal
200g urea * 1 mole urea * 1 mole urea methanal = 3.33 mole urea methanal
60.06 g urea 1 mole urea
Conclusion:
Urea is the limiting reactant in this reaction since it produced the least product which is urea methanal.
References
http://chemwiki.ucdavis.edu/Physical_Chemistry/Physical_Properties_of_Matter/Atomic_and_Molecular_Properties/Intermolecular_Forces/Intermolecular_Forces
Urea in car engines :
http://www.discoverdef.com/def-overview/faq/#def
http://www.wisegeek.com/what-is-the-use-of-urea-in-diesel.htm
Urea Cycle:
http://www.slideshare.net/ednapeter5/urea-cycle-14919824
Pictures:
http://s2.hubimg.com/u/1298189_f520.jpg
http://en.wikipedia.org/wiki/Urea_cycle
Urea Nitrate:
http://www.wisegeek.com/what-is-urea-nitrate.htm
http://en.wikipedia.org/wiki/Urea_nitrate
Reactions:
http://www.chemspider.com/Chemical-Structure.1143.html
http://www.uq.edu.au/_School_Science_Lessons/appendixD.html
Videos:
Urea Cycle: https://www.youtube.com/watch?v=-5aSLN9LWCE
Explosion: https://www.youtube.com/watch?v=5SpTofaUY0I
Urea in car engines :
http://www.discoverdef.com/def-overview/faq/#def
http://www.wisegeek.com/what-is-the-use-of-urea-in-diesel.htm
Urea Cycle:
http://www.slideshare.net/ednapeter5/urea-cycle-14919824
Pictures:
http://s2.hubimg.com/u/1298189_f520.jpg
http://en.wikipedia.org/wiki/Urea_cycle
Urea Nitrate:
http://www.wisegeek.com/what-is-urea-nitrate.htm
http://en.wikipedia.org/wiki/Urea_nitrate
Reactions:
http://www.chemspider.com/Chemical-Structure.1143.html
http://www.uq.edu.au/_School_Science_Lessons/appendixD.html
Videos:
Urea Cycle: https://www.youtube.com/watch?v=-5aSLN9LWCE
Explosion: https://www.youtube.com/watch?v=5SpTofaUY0I